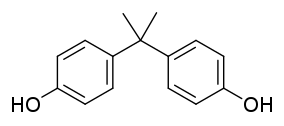
Bisphenol A
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4,4′-(Propane-2,2-diyl)diphenol | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.001.133 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2924 2430 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H16O2 | |
| Molar mass | 228.291 g·mol−1 |
| Appearance | White solid |
| Density | 1.20 g/cm3 |
| Melting point | 155 °C (311 °F; 428 K)[1] |
| Boiling point | 250–252 °C (482–486 °F; 523–525 K)[1] at 13 torrs (0.017 atm) |
| 120–300 ppm (21.5 °C) | |
| Vapor pressure | 5×10−6 Pa (25 °C)[2] |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H317, H318, H335, H360 | |
| P201, P202, P261, P271, P272, P280, P281, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P312, P321, P333+P313, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 227 °C (441 °F; 500 K) |
| 600 °C (1,112 °F; 873 K) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Bisphenol A (BPA) is a chemical compound primarily used in the manufacture of various plastics. It is a colourless solid which is soluble in organic solvents, but is only poorly soluble in water.[3] It is produced on an industrial scale by the condensation of phenol and acetone, with global production in 2022 expected to reach 10 million tonnes.[4]
Its largest single application is as a co-monomer in the production of polycarbonates, which accounts for 65-70% of all BPA production.[5][6] 25-30% is used in the manufacture of epoxy resins and vinyl ester resins.[5][6] The remaining 5% is used as a major component of several high-performance plastics and as a minor additive in PVC, polyurethane, thermal paper and several other materials. It is not a plasticizer,[7] although it is often wrongly labelled as such.
The heath effects of BPA have been the subject of prolonged public and scientific debate.[8][9][10] BPA is a xenoestrogen, exhibiting estrogen-mimicking, hormone-like properties.[11] Although the effect is very weak, the pervasiveness of BPA-containing materials raises concerns, as exposure is effectively lifelong. Many BPA-containing materials are non-obvious but commonly encountered,[12] and include coatings for the inside of food cans,[13] clothing,[14] shop receipts[15] and dental fillings.[16] BPA has been investigated by public health agencies in many countries, as well as by the World Health Organization.[8] While normal exposure is below the level currently associated with risk, several jurisdictions have taken steps to reduce exposure on a precautionary basis, in particular by banning BPA from baby bottles. There is some evidence that BPA exposure in infants has decreased as a result of this.[17] BPA-free plastics have also been introduced, which are manufactured using alternative bisphenols such as bisphenol S and bisphenol F, but there is controversy around whether these are actually safer.[18]
History
Bisphenol A was reported in 1891 by the Russian chemist Aleksandr Dianin.[19]
In 1934 workers at I.G. Farbenindustrie reported the coupling of BPA and epichlorohydrin. Over the following decade, coatings and resins derived from similar materials were described by workers at the companies of DeTrey Freres in Switzerland and DeVoe and Raynolds in the US. This early work underpinned the development of epoxy resins, which in turn motivated production of BPA.[20] The utilization of BPA further expanded with discoveries at Bayer and General Electric on polycarbonate plastics. These plastics first appeared in 1958, being produced by Mobay and General Electric, and Bayer.[21]
In terms of the endocrine disruption controversy, the British biochemist Edward Charles Dodds tested BPA as an artificial estrogen in the early 1930s. He found BPA to be 1 / 37,000 as effective as estradiol.[22][23][24] Dodds eventually developed a structurally similar compound, diethylstilbestrol (DES), which was used as a synthetic estrogen drug in women and animals until it was banned due to its risk of causing cancer; the ban on use of DES in humans came in 1971 and in animals, in 1979.[22] BPA was never used as a drug.[22]
Production
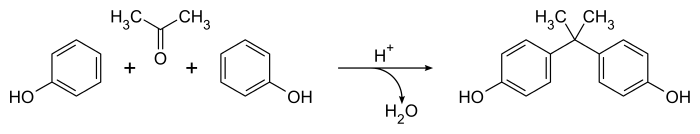
The synthesis of BPA still follows Dianin's general method, with the fundamentals having changed little in 130 years. The condensation of acetone (hence the suffix 'A' in the name)[25] with two equivalents of phenol is catalyzed by a strong acid, such as concentrated hydrochloric acid, sulfuric acid, or a solid acid resin such as polystyrene sulfonate.[26] An excess of phenol is used to ensure full condensation and to limit the formation of by-products, such as Dianin's compound. Large amounts of both starting materials are available from the cumene process,[3] making it fairly cheap to produce. Global production in 2022 is expected to reach 10 million tonnes.[4]
The reaction is strongly para selective but minor amounts of the ortho-para (up 3%) and ortho-ortho isomers are also produced, along with several other minor by-products.[27] These are not always removed and are known impurities in commercial samples of BPA.[28][27]
Uses and applications

Polycarbonates
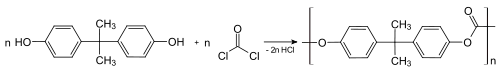
Between about 65-70% of all bisphenol A is used to make polycarbonate plastics,[5][6] which can consists of nearly 90% BPA by mass. Polymerisation is achieved by a reaction with phosgene, conducted under biphasic conditions; the hydrochloric acid is scavenged with aqueous base.[29] This process converts the individual molecules of BPA into large polymer chains, effectively trapping them.
Epoxy and vinyl ester resins
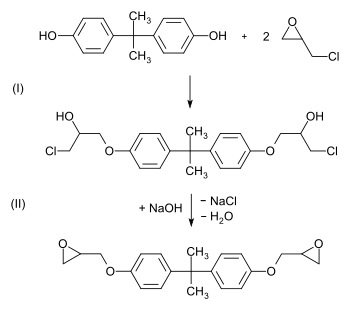
About 25-30% of all BPA is used in the manufacture of epoxy resins and vinyl ester resins.[5][6] For epoxy resin it is first converted to its diglycide ether (usually abbreviated BADGE or DGEBA).[30][31] This is achieved by a reaction with epichlorohydrin under basic conditions.
Some of this is further reacted with methacrylic acid to form bis-GMA, which is used to make vinyl ester resins. Alternatively, and to a much lesser extent, BPA may be ethoxylated and then converted to its diacrylate and dimethacrylate derivatives. These may be incorporated at low levels in vinyl ester resins to change their physical properties.[32]
Minor uses
The remaining 5% of BPA is used in a wide range of applications, many of which are also to do with plastic.[33] BPA is a major component of several high-performance plastics, the production of these is low compared to other plastics but still equals several thousand tons a year. Comparatively minor amounts of BPA are also used as additives or modifiers in some commodity plastics. These materials are much more common but their BPA content will be minimal.
Plastics
- As a major component
- Polycyanurates can be produced from BPA by way of its cyanate ester.[33] This is itself formed by a reaction between BPA and cyanogen bromide. Examples include BT-Epoxy, which is one of a number of resins used the production of printed circuit boards.
- Polyetherimides can be produced from BPA.[34] These plastics have exceptional resistance to mechanical, thermal and chemical damage. They find use in medical devices and other high performance instrumentation.
- Polybenzoxazines may be produced from a number of biphenols, including BPA.[35][36]
- Polysulfones can be produced from BPA and bis(4-chlorophenyl) sulfone forming poly(bisphenol-A sulfone) (PSF). It is used as a high performance alternative to polycarbonate.[33]
- Bisphenol-A formaldehyde resins are a subset of phenol formaldehyde resins. They are used in the production of high-pressure laminates[33]
- As a minor component
- Polyurethane foams, and particularly memory foams, sometimes use the diacrylate and dimethacrylate derivatives of ethoxylated BPA as chain extenders.[37]
- PVC can contain BPA and its derivatives through multiple routes. BPA is sometimes used as an antioxidant in phthalates,[38] which are extensively used as plasticizers for PVC. BPA has also been used as an antioxidant to protect sensitive PVC heat stabilizers. Historically between 5 and 10% by weight of BPA was included in barium cadmium types, and from 5 to 10% of BHT was included in nontoxic calcium zinc solids. BPA diglycidyl ether (BADGE) is used as an acid scavenger, particularly in PVC dispersions, such as organosols or plastisols,[39][40] which are used as coatings for the inside of food cans, as well as embossed clothes designs produced using heat transfer vinyl or screen printing machines.[14]
- Bromination of BPA froms tetrabromobisphenol A (TBBPA), which is used as a flame retardant in plastics.[41] TBBPA is mainly used as a reactive component of polymers, meaning that it is incorporated into the polymer backbone. It is used to prepare fire-resistant polycarbonates by replacing some bisphenol A. A lower grade of TBBPA is used to prepare epoxy resins, used in printed circuit boards. Its use is diminishing due to restrictions on brominated flame retardants.
Other applications
- BPA finds use as an antioxidant in several fields, partiually in brake fluids.[42]
- BPA is used as a developering agent in thermal paper (shop receipts).[15] Recycled paper products can also contain BPA as a result,[43] although this can depend strongly on how it is recycled. Deinking can remove 95% of BPA,[5] with the pulp produced used to make newsprint, toilet paper and facial tissues. If deinking is not performed then the BPA remains in the fibres, paper recycled this way is usually made into corrugated fiberboard.[5]
- Ethoxylated BPA finds minor use as a 'levelling agent' in tin electroplating.
- Several drug candidates have also been developed from bisphenol A, including Ralaniten, Ralaniten acetate, and EPI-001.
BPA substitutes
Concerns about the health effects of BPA have led many manufacturers to replace BPA with substitutes, particularly bisphenol S and bisphenol F. However, health concerns have been raised about these substitutes as well.[44] In the similar compound Bisphenol F, the F signifies formaldehyde Numerous ketones undergo analogous condensation reactions.[3]
| Structural formula | Name | CAS | Reactants | |
|---|---|---|---|---|
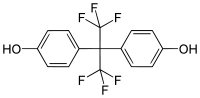 | Bisphenol AF | 1478-61-1 | Phenol | Hexafluoroacetone |
| Bisphenol F | 620-92-8 | Phenol | Formaldehyde | |
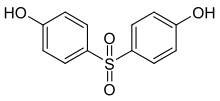 | Bisphenol S | 80-09-1 | Phenol | Sulfur trioxide |
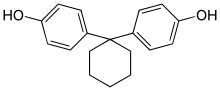 | Bisphenol Z | 843-55-0 | Phenol | Cyclohexanone |
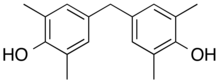 | Tetramethyl bisphenol F | 5384-21-4 | 2,6-xylenol | Formaldehyde |
Human safety
Exposure

The presence of BPA in plastics and other commonplace materials means that most people are frequently exposed to trace levels of this compound.[45][46][47] The primary source of human exposure is via food, as epoxy and PVC are used to line the inside of food cans to prevent corrosion of the metal by acidic foodstuffs. Polycarbonate drinks containers are also a source of exposure, although most disposable drinks bottles are actually made of PET, which contains no BPA. Among the non-food sources, exposures routes include through dust,[6] thermal paper,[15] clothing,[14] dental materials, and medical devices.[12] Although BPA exposure is common it does not accumulate within the body, with toxicokinetic studies show the biological half-life of BPA in adult humans to be around 2 hours.[48][49] The body first converts it into more water soluble compounds via glucuronidation or sulfation, which are then removed in urine. This allows for total exposure to be easily motioned by urine testing.[17][12]
Health effects and regulation
The heath effects of BPA have been the subject of prolonged public and scientific debate.[8][9][10] Concern is mostly related to its estrogen-like activity, although it can interact with other receptor systems as an endocrine-disrupting chemical. These interactions are all very weak, but exposure to BPA is effectively lifelong, leading to concern over possible cumulative effects. Studying this sort of long-term, low-dose interaction is difficult, and although there have been numerous studies, there are considerable discrepancies in their conclusions regarding the nature of the effects observed as well as the levels at which they occur.[8] A common criticism is that industry-sponsored trials tend to show BPA as being safer than studies performed by academic or government laboratories,[10][50] although this has also been explained in terms of industry studies being better designed.[9]
Public health agencies in the EU,[51][52] US,[53][54] Canada,[55] Australia[56] and Japan as well as the WHO[8] have all reviewed the health risks of BPA, and found normal exposure to be below the level currently associated with risk. Regardless, due to the scientific uncertainty, many jurisdictions have taken steps to reduce exposure on a precautionary basis. In particular, infants are considered to be at greater risk,[57] leading to bans on the use of BPA in baby bottles and related products by the US,[58] Canada,[59] and EU[60] amongst others. Bottle producers have largely switch from polycarbonate to polypropylene and there is some evidence that BPA exposure in infants has decreased as a result of this.[17] The European Chemicals Agency has added BPA to the Candidate List of substances of very high concern (SVHC), which would make it easier to restrict or ban its use in future.[61][62] In the United States, there are 12 states, in addition to Washington, D.C., that have restrictions in place against BPA in some way.[63]
Toxicity
BPA exhibits very low acute toxicity as indicated by its LD50 of 4 g/kg (mouse). In those mice, weight gain was reduced and exhibited estrogen-like properties. Reports indicate that it is a minor skin irritator as well, although less so than phenol.[3]
Pharmacology
BPA has been found to bind to both of the nuclear estrogen receptors (ERs), ERα and ERβ. It is 1000- to 2000-fold less potent than estradiol. BPA can both mimic the action of estrogen and antagonize estrogen, indicating that it is a selective estrogen receptor modulator (SERM) or partial agonist of the ER. At high concentrations, BPA also binds to and acts as an antagonist of the androgen receptor (AR). In addition to receptor binding, the compound has been found to affect Leydig cell steroidogenesis, including affecting 17α-hydroxylase/17,20 lyase and aromatase expression and interfering with LH receptor-ligand binding.
In 1997, adverse effects of low-dose BPA exposure in laboratory animals were first proposed.[64] Some studies have found that BPA increases anxiety in rats.[65][66] Modern studies began finding possible connections to health issues caused by exposure to BPA during pregnancy and during development. As of 2014, research and debates are ongoing as to whether BPA should be banned or not.
A 2007 study investigated the interaction between bisphenol A's and estrogen-related receptor γ (ERR-γ). This orphan receptor (endogenous ligand unknown) behaves as a constitutive activator of transcription. BPA seems to bind strongly to ERR-γ (dissociation constant = 5.5 nM), but only weakly to the ER.[67] BPA binding to ERR-γ preserves its basal constitutive activity.[67] It can also protect it from deactivation from the SERM 4-hydroxytamoxifen (afimoxifene).[67] This may be the mechanism by which BPA acts as a xenoestrogen.[67] Different expression of ERR-γ in different parts of the body may account for variations in bisphenol A effects. BPA has also been found to act as an agonist of the GPER (GPR30).[68]
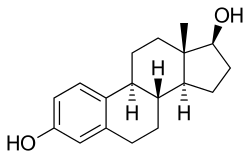 Estradiol: the major female sex hormone in humans
Estradiol: the major female sex hormone in humans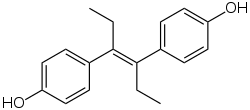 Diethylstilbestrol: a powerful synthetic estrogen
Diethylstilbestrol: a powerful synthetic estrogen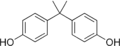 Bisphenol A
Bisphenol A
Environmental safety
Prevalence and persistence
BPA has been detectable in the natural environment since the 1990s and is now widely distributed.[69] It is primarily a river pollutant,[70] but has also been observed in the marine environment,[71] and in soils.[72] Lower levels can also been detected in air.[73] The solubility of BPA in water is low (~300 g/ton of water) but this is still sufficient to make it a significant means of distribution into the environment.[72] Many of the largest sources of BPA pollution are water-based, particularly wastewater from industrial facilities using BPA. Paper recycling can be a major source of release when this includes thermal paper,[5][74] leaching from PVC items may also be a significant source,[70] as can landfill leachate.[75]
In all cases wastewater treatment can be highly effective at removing BPA, with reductions of 91–98%.[76] Regardless, the remaining 2-9% of BPA will continue through to the environment, with low levels of BPA commonly observed in surface water and sediment in the U.S. and Europe.[77]
Once in the environment BPA is aerobically biodegraded by a wide a variety of organisms.[69][78][79] It's half life in water has been estimated at between 4.5 - 15 days, degradation in the air is faster than this, while soil samples degrade more slowly.[72] BPA in sediment degrades most slowly of all, particularly where this is anaerobic.
Environmental effects
BPA is environmental contaminant of emerging concern.[75] Despite its short half-life and non-bioaccumulating character, the continuous release of BPA into the environment causes continuous exposure to both animal and plant life.[80] Although many studies have been performed, these often focus on a limited range of model organisms and can use BPA concentrations well beyond environmental levels.[81] As such, the precise effects of BPA on the growth, reproduction, and development of aquatic organism is not fully understood.[81] Regardless, the existing data shows the effects of BPA on wildlife to be generally negative.[82][83] Certain species, such as invertebrates and amphibians seem to be particularly affected[82] BPA also appears to effect development and reproduction in a wide range of wildlife species.[83]
See also
References
- Mitrofanova, S. E.; Bakirova, I. N.; Zenitova, L. A.; Galimzyanova, A. R.; Nefed’ev, E. S. (September 2009). "Polyurethane varnish materials based on diphenylolpropane". Russian Journal of Applied Chemistry. 82 (9): 1630–1635. doi:10.1134/S1070427209090225.
- "Chemical Fact Sheet – Cas #80057 CASRN 80-05-7". speclab.com. 1 April 2012. Archived from the original on 12 February 2012. Retrieved 14 June 2012.
- Fiege H, Voges HW, Hamamoto T, Umemura S, Iwata T, Miki H, Fujita Y, Buysch HJ, Garbe D, Paulus W (2000). "Phenol Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313.
- Abraham A, Chakraborty P (June 2020). "A review on sources and health impacts of bisphenol A". Reviews on Environmental Health. 35 (2): 201–210. doi:10.1515/reveh-2019-0034. PMID 31743105. S2CID 208186123.
- European Commission. Joint Research Centre. Institute for Health Consumer Protection (2010). Updated European Union risk assessment report : 4,4'-isopropylidenediphenol (bisphenol-A) : environment addendum of February 2008. Publications Office. p. 6. doi:10.2788/40195. ISBN 9789279175411.
- Vasiljevic T, Harner T (May 2021). "Bisphenol A and its analogues in outdoor and indoor air: Properties, sources and global levels". The Science of the Total Environment. 789: 148013. Bibcode:2021ScTEn.789n8013V. doi:10.1016/j.scitotenv.2021.148013. PMID 34323825.
- Cadogan DF, Howick CJ (2000). "Plasticizers". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a20_439. ISBN 3527306730.
- Joint FAO/WHO expert meeting to review toxicological and health aspects of bisphenol A : final report, including report of stakeholder meeting on bisphenol A, 1-5 November 2010, Ottawa, Canada. World Health Organization. 2011. hdl:10665/44624. ISBN 97892141564274. Retrieved 23 March 2022.
{{cite book}}: Check|isbn=value: length (help) - Hengstler JG, Foth H, Gebel T, Kramer PJ, Lilienblum W, Schweinfurth H, et al. (April 2011). "Critical evaluation of key evidence on the human health hazards of exposure to bisphenol A". Critical Reviews in Toxicology. 41 (4): 263–291. doi:10.3109/10408444.2011.558487. PMC 3135059. PMID 21438738.
- Myers JP, vom Saal FS, Akingbemi BT, Arizono K, Belcher S, Colborn T, et al. (March 2009). "Why public health agencies cannot depend on good laboratory practices as a criterion for selecting data: the case of bisphenol A". Environmental Health Perspectives. 117 (3): 309–315. doi:10.1289/ehp.0800173. PMC 2661896. PMID 19337501.
- Egan M (2013). "Sarah A. Vogel. Is It Safe? BPA and the Struggle to Define the Safety of Chemicals". Isis. Berkeley: University of California Press. 105 (1): 254. doi:10.1086/676809. ISSN 0021-1753.
- Geens T, Aerts D, Berthot C, Bourguignon JP, Goeyens L, Lecomte P, et al. (October 2012). "A review of dietary and non-dietary exposure to bisphenol-A". Food and Chemical Toxicology. 50 (10): 3725–3740. doi:10.1016/j.fct.2012.07.059. PMID 22889897.
- Noonan GO, Ackerman LK, Begley TH (July 2011). "Concentration of bisphenol A in highly consumed canned foods on the U.S. market". Journal of Agricultural and Food Chemistry. 59 (13): 7178–7185. doi:10.1021/jf201076f. PMID 21598963.
- Xue J, Liu W, Kannan K (May 2017). "Bisphenols, Benzophenones, and Bisphenol A Diglycidyl Ethers in Textiles and Infant Clothing". Environmental Science & Technology. 51 (9): 5279–5286. Bibcode:2017EnST...51.5279X. doi:10.1021/acs.est.7b00701. PMID 28368574.
- Björnsdotter MK, de Boer J, Ballesteros-Gómez A (September 2017). "Bisphenol A and replacements in thermal paper: A review". Chemosphere. 182: 691–706. Bibcode:2017Chmsp.182..691B. doi:10.1016/j.chemosphere.2017.05.070. PMID 28528315.
- Ahovuo-Saloranta A, Forss H, Walsh T, Nordblad A, Mäkelä M, Worthington HV (July 2017). "Pit and fissure sealants for preventing dental decay in permanent teeth". The Cochrane Database of Systematic Reviews. 7: CD001830. doi:10.1002/14651858.CD001830.pub5. PMC 6483295. PMID 28759120.
- Huang RP, Liu ZH, Yin H, Dang Z, Wu PX, Zhu NW, Lin Z (June 2018). "Bisphenol A concentrations in human urine, human intakes across six continents, and annual trends of average intakes in adult and child populations worldwide: A thorough literature review". The Science of the Total Environment. 626: 971–981. Bibcode:2018ScTEn.626..971H. doi:10.1016/j.scitotenv.2018.01.144. PMID 29898562. S2CID 49194096.
- Thoene M, Dzika E, Gonkowski S, Wojtkiewicz J (February 2020). "Bisphenol S in Food Causes Hormonal and Obesogenic Effects Comparable to or Worse than Bisphenol A: A Literature Review". Nutrients. 12 (2): 532. doi:10.3390/nu12020532. PMC 7071457. PMID 32092919.
- See:
- А. Дианина (1891) "О продуктахъ конденсацiи кетоновъ съ фенолами" (On condensation products of ketones with phenols), Журнал Русского физико-химического общества (Journal of the Russian Physical Chemistry Society), 23 : 488-517, 523–546, 601–611; see especially pages 491-493 ("Диметилдифенолметань" (dimethyldiphenolmethane)).
- Reprinted in condensed form in: A. Dianin (1892) "Condensationsproducte aus Ketonen und Phenolen" (Condensation products of ketones and phenols), Berichte der Deutschen chemischen Gesellschaft zu Berlin, 25, part 3 : 334-337.
- Pham HQ, Marks MJ (2012). Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_547.pub2.
- Serini V (2000). Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_207.
- Vogel SA (November 2009). "The politics of plastics: the making and unmaking of bisphenol a "safety"". American Journal of Public Health. 99 (Suppl 3): S559–S566. doi:10.2105/AJPH.2008.159228. PMC 2774166. PMID 19890158.
- Dodds EC, Lawson W (1936). "Synthetic Œstrogenic Agents without the Phenanthrene Nucleus". Nature. 137 (3476): 996. Bibcode:1936Natur.137..996D. doi:10.1038/137996a0. S2CID 4171635.
- Dodds EC, Lawson W (1938). "Molecular Structure in Relation to Oestrogenic Activity. Compounds without a Phenanthrene Nucleus". Proceedings of the Royal Society of London B: Biological Sciences. 125 (839): 222–232. Bibcode:1938RSPSB.125..222D. doi:10.1098/rspb.1938.0023.
- Uglea CV, Negulescu II (1991). Synthesis and Characterization of Oligomers. CRC Press. p. 103. ISBN 978-0-8493-4954-6.
- De Angelis A, Ingallina P, Perego C (March 2004). "Solid Acid Catalysts for Industrial Condensations of Ketones and Aldehydes with Aromatics". Industrial & Engineering Chemistry Research. 43 (5): 1169–1178. doi:10.1021/ie030429+.
- Terasaki M, Nomachi M, Edmonds JS, Morita M (May 2004). "Impurities in industrial grade 4,4'-isopropylidene diphenol (bisphenol A): possible implications for estrogenic activity". Chemosphere. 55 (6): 927–931. Bibcode:2004Chmsp..55..927T. doi:10.1016/j.chemosphere.2003.11.063. PMID 15041297.
- Pahigian JM, Zuo Y (September 2018). "Occurrence, endocrine-related bioeffects and fate of bisphenol A chemical degradation intermediates and impurities: A review". Chemosphere. 207: 469–480. Bibcode:2018Chmsp.207..469P. doi:10.1016/j.chemosphere.2018.05.117. PMID 29807346. S2CID 44172964.
- Serini V (2000). "Polycarbonates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_207.
- Ng F, Couture G, Philippe C, Boutevin B, Caillol S (January 2017). "Bio-Based Aromatic Epoxy Monomers for Thermoset Materials". Molecules. 22 (1): 149. doi:10.3390/molecules22010149. PMC 6155700. PMID 28106795.
- Kroschwitz JI (1998). Kirk-Othmer Encyclopedia of Chemical Technology. Vol. 5 (5 ed.). p. 8. ISBN 978-0-471-52695-7.
- Gonçalves F, Kawano Y, Pfeifer C, Stansbury JW, Braga RR (August 2009). "Influence of BisGMA, TEGDMA, and BisEMA contents on viscosity, conversion, and flexural strength of experimental resins and composites". European Journal of Oral Sciences. 117 (4): 442–446. doi:10.1111/j.1600-0722.2009.00636.x. PMID 19627357.
- Geens T, Goeyens L, Covaci A (September 2011). "Are potential sources for human exposure to bisphenol-A overlooked?". International Journal of Hygiene and Environmental Health. 214 (5): 339–347. doi:10.1016/j.ijheh.2011.04.005. PMID 21570349.
- Takekoshi T, Kochanowski JE, Manello JS, Webber MJ (June 1985). "Polyetherimides. I. Preparation of dianhydrides containing aromatic ether groups". Journal of Polymer Science: Polymer Chemistry Edition. 23 (6): 1759–1769. Bibcode:1985JPoSA..23.1759T. doi:10.1002/pol.1985.170230616.
- Vijayakumar CT, Shamim Rishwana S, Surender R, David Mathan N, Vinayagamoorthi S, Alam S (2 January 2014). "Structurally diverse benzoxazines: synthesis, polymerization, and thermal stability". Designed Monomers and Polymers. 17 (1): 47–57. doi:10.1080/15685551.2013.797216. S2CID 94255723.
- Ghosh NN, Kiskan B, Yagci Y (November 2007). "Polybenzoxazines—New high performance thermosetting resins: Synthesis and properties". Progress in Polymer Science. 32 (11): 1344–1391. doi:10.1016/j.progpolymsci.2007.07.002.
- Laza JM, Veloso A, Vilas JL (10 January 2021). "Tailoring new bisphenol a ethoxylated shape memory polyurethanes". Journal of Applied Polymer Science. 138 (2): 49660. doi:10.1002/app.49660. S2CID 224955435.
- "European Union Summary Risk Assessment Report - Bis (2-ethylhexyl) Phthalate (DEHP)". Joint Research Centre (JRC) Publications Repository. European Commission. 16 July 2008. ISSN 1018-5593. Retrieved 24 November 2021.

- Shah AC, Poledna DJ (September 2003). "Review of PVC dispersion and blending resin products". Journal of Vinyl and Additive Technology. 9 (3): 146–154. doi:10.1002/vnl.10076. S2CID 98016356.
- Shah AC, Poledna DJ (September 2002). "Review of specialty PVC resins". Journal of Vinyl and Additive Technology. 8 (3): 214–221. doi:10.1002/vnl.10365. S2CID 97146596.
- Dagani MJ, Barda HJ, Benya TJ, Sanders DC. "Bromine Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a04_405.
- Lamprea K, Bressy A, Mirande-Bret C, Caupos E, Gromaire MC (August 2018). "Alkylphenol and bisphenol A contamination of urban runoff: an evaluation of the emission potentials of various construction materials and automotive supplies". Environmental Science and Pollution Research International. 25 (22): 21887–21900. doi:10.1007/s11356-018-2272-z. PMID 29796891. S2CID 44140721.
- Liao C, Kannan K (November 2011). "Widespread occurrence of bisphenol A in paper and paper products: implications for human exposure". Environmental Science & Technology. 45 (21): 9372–9379. Bibcode:2011EnST...45.9372L. doi:10.1021/es202507f. PMID 21939283.
- Rochester JR, Bolden AL (July 2015). "Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes". Environmental Health Perspectives. 123 (7): 643–650. doi:10.1289/ehp.1408989. PMC 4492270. PMID 25775505.
- Calafat AM, Ye X, Wong LY, Reidy JA, Needham LL (January 2008). "Exposure of the U.S. population to bisphenol A and 4-tertiary-octylphenol: 2003-2004". Environmental Health Perspectives. 116 (1): 39–44. doi:10.1289/ehp.10753. PMC 2199288. PMID 18197297.
- Thoene M, Rytel L, Nowicka N, Wojtkiewicz J (May 2018). "The state of bisphenol research in the lesser developed countries of the EU: a mini-review". Toxicology Research. 7 (3): 371–380. doi:10.1039/c8tx00064f. PMC 6062254. PMID 30090587.
- Vandenberg LN, Hauser R, Marcus M, Olea N, Welshons WV (August 2007). "Human exposure to bisphenol A (BPA)". Reproductive Toxicology. 24 (2): 139–177. doi:10.1016/j.reprotox.2007.07.010. PMID 17825522.
- Tsukioka T, Terasawa JI, Sato S, Hatayama Y, Makino T, Nakazawa H (2004). "Development of Analytical Method for Determining Trace Amounts of BPA in Urine Samples and Estimation of Exposure to BPA". Journal of Environmental Chemistry. 14 (1): 57–63. doi:10.5985/jec.14.57.
- Shin BS, Kim CH, Jun YS, Kim DH, Lee BM, Yoon CH, et al. (December 2004). "Physiologically based pharmacokinetics of bisphenol A". Journal of Toxicology and Environmental Health. Part A. 67 (23–24): 1971–1985. doi:10.1080/15287390490514615. PMID 15513896. S2CID 24467830.
- vom Saal FS, Hughes C (2005). "An extensive new literature concerning low-dose effects of bisphenol A shows the need for a new risk assessment". Environ. Health Perspect. 113 (8): 926–33. doi:10.1289/ehp.7713. PMC 1280330. PMID 16079060.
- "Bisphenol A - ECHA". echa.europa.eu. Retrieved 28 March 2022.
- European Food Safety Authority (2015). EFSA explains the Safety of Bisphenol A: scientific opinion on bisphenol A (2015). European Food Safety Authority. doi:10.2805/075460. ISBN 9789291996421.
- OCSPP US EPA (21 September 2015). "Risk Management for Bisphenol A (BPA)". www.epa.gov. Retrieved 28 March 2022.
- CLARITY-BPA Research Program (October 2021). NTP Research Report on the Consortium Linking Academic and Regulatory Insights on Bisphenol A Toxicity (CLARITY-BPA): A Compendium of Published Findings. p. 18. doi:10.22427/NTP-RR-18. PMID 34910417. S2CID 240266384.
- Health Canada (16 April 2013). "Bisphenol A (BPA)". www.canada.ca. Retrieved 28 March 2022.
- "Bisphenol A (BPA)". www.foodstandards.gov.au. Retrieved 28 March 2022.
- Aschberger K, Castello P, Hoekstra E (2010). "Bisphenol A and baby bottles : challenges and perspectives". The Publications Office of the European Union. doi:10.2788/97553. ISBN 9789279158698.
- "Indirect Food Additives: Polymers". Federal Register. U.S. Government Publishing Office.77 FR 41899
- Legislative Services Branch (1 July 2020). "Consolidated federal laws of canada, Canada Consumer Product Safety Act". laws-lois.justice.gc.ca.
- "EUR-Lex - 32011L0008 - EN - EUR-Lex". EUR-Lex. European Union.
COMMISSION DIRECTIVE 2011/8/EU of 28 January 2011 amending Directive 2002/72/EC as regards the restriction of use of Bisphenol A in plastic infant feeding bottles
- "MSC unanimously agrees that Bisphenol A is an endocrine disruptor - All news - ECHA". echa.europa.eu. Retrieved 19 June 2017.
- Fisher D (12 July 2019). "EU court confirms BPA as substance of 'very high concern'". Environmental Health News. Retrieved 21 July 2020.
- "NCSL Policy Update: State Restrictions on Bisphenol A (BPA) in Consumer Products". NCSL.org.
- Erickson BE (2 June 2008). "Bisphenol A under scrutiny". Chemical and Engineering News. 86 (22): 36–39. doi:10.1021/cen-v086n022.p036.
- Hicks KD, Sullivan AW, Cao J, Sluzas E, Rebuli M, Patisaul HB (August 2016). "Interaction of bisphenol A (BPA) and soy phytoestrogens on sexually dimorphic sociosexual behaviors in male and female rats". Hormones and Behavior. 84: 121–126. doi:10.1016/j.yhbeh.2016.06.010. PMC 4996731. PMID 27373758.
- Shipman M (7 September 2012). "Study Finds How BPA Affects Gene Expression, Anxiety; Soy Mitigates Effects". NC State News. Retrieved 4 February 2021.
- Matsushima A, Kakuta Y, Teramoto T, Koshiba T, Liu X, Okada H, et al. (October 2007). "Structural evidence for endocrine disruptor bisphenol A binding to human nuclear receptor ERR gamma". Journal of Biochemistry. 142 (4): 517–524. doi:10.1093/jb/mvm158. PMID 17761695.
- Prossnitz ER, Barton M (May 2014). "Estrogen biology: new insights into GPER function and clinical opportunities". Molecular and Cellular Endocrinology. 389 (1–2): 71–83. doi:10.1016/j.mce.2014.02.002. PMC 4040308. PMID 24530924.
- Staples, Charles A.; Dome, Philip B.; Klecka, Gary M.; Oblock, Sondra T.; Harris, Lynne R. (April 1998). "A review of the environmental fate, effects, and exposures of bisphenol A". Chemosphere. 36 (10): 2149–2173. Bibcode:1998Chmsp..36.2149S. doi:10.1016/S0045-6535(97)10133-3. PMID 9566294.
- Corrales J, Kristofco LA, Steele WB, Yates BS, Breed CS, Williams ES, Brooks BW (29 July 2015). "Global Assessment of Bisphenol A in the Environment: Review and Analysis of Its Occurrence and Bioaccumulation". Dose-Response. 13 (3): 1559325815598308. doi:10.1177/1559325815598308. PMC 4674187. PMID 26674671.
- Ozhan, Koray; Kocaman, Emel (February 2019). "Temporal and Spatial Distributions of Bisphenol A in Marine and Freshwaters in Turkey". Archives of Environmental Contamination and Toxicology. 76 (2): 246–254. doi:10.1007/s00244-018-00594-6. PMID 30610254. S2CID 58536418.
- Cousins, I.T.; Staples, C.A.; Kleĉka, G.M.; Mackay, D. (July 2002). "A Multimedia Assessment of the Environmental Fate of Bisphenol A". Human and Ecological Risk Assessment: An International Journal. 8 (5): 1107–1135. doi:10.1080/1080-700291905846. S2CID 43509780.
- Vasiljevic, Tijana; Harner, Tom (October 2021). "Bisphenol A and its analogues in outdoor and indoor air: Properties, sources and global levels". Science of the Total Environment. 789: 148013. Bibcode:2021ScTEn.789n8013V. doi:10.1016/j.scitotenv.2021.148013. PMID 34323825.
- Fürhacker, M; Scharf, S; Weber, H (September 2000). "Bisphenol A: emissions from point sources". Chemosphere. 41 (5): 751–756. Bibcode:2000Chmsp..41..751F. doi:10.1016/S0045-6535(99)00466-X. PMID 10834378.
- Qi, Chengdu; Huang, Jun; Wang, Bin; Deng, Shubo; Wang, Yujue; Yu, Gang (2018). "Contaminants of emerging concern in landfill leachate in China: A review". Emerging Contaminants. 4 (1): 1–10. doi:10.1016/j.emcon.2018.06.001.
- Drewes JE, Hemming J, Ladenburger SJ, Schauer J, Sonzogni W (2005). "An assessment of endocrine disrupting activity changes during wastewater treatment through the use of bioassays and chemical measurements". Water Environment Research. 77 (1): 12–23. doi:10.2175/106143005x41573. PMID 15765931. S2CID 12283834.
- Klecka GM, Staples CA, Clark KE, Van der Hoeven N, Thomas DE, Hentges SG (August 2009). "Exposure analysis of bisphenol A in surface water systems in North America and Europe". Environmental Science & Technology. 43 (16): 6145–50. Bibcode:2009EnST...43.6145K. doi:10.1021/es900598e. PMID 19746705.
- Kang, J; Katayama, Y; Kondo, F (16 January 2006). "Biodegradation or metabolism of bisphenol A: From microorganisms to mammals". Toxicology. 217 (2–3): 81–90. doi:10.1016/j.tox.2005.10.001. PMID 16288945.
- Zhang, Chi; Li, Yi; Wang, Chao; Niu, Lihua; Cai, Wei (2 January 2016). "Occurrence of endocrine disrupting compounds in aqueous environment and their bacterial degradation: A review". Critical Reviews in Environmental Science and Technology. 46 (1): 1–59. doi:10.1080/10643389.2015.1061881. S2CID 94353391.
- Xiao, Changyun; Wang, Lihong; Zhou, Qing; Huang, Xiaohua (February 2020). "Hazards of bisphenol A (BPA) exposure: A systematic review of plant toxicology studies". Journal of Hazardous Materials. 384: 121488. doi:10.1016/j.jhazmat.2019.121488. PMID 31699483. S2CID 207939269.
- Rubin, Alexander M.; Seebacher, Frank (July 2022). "Bisphenols impact hormone levels in animals: A meta-analysis". Science of the Total Environment. 828: 154533. Bibcode:2022ScTEn.828o4533R. doi:10.1016/j.scitotenv.2022.154533. PMID 35288143. S2CID 247423338.
- Wu, Nicholas C.; Seebacher, Frank (July 2020). "Effect of the plastic pollutant bisphenol A on the biology of aquatic organisms: A meta‐analysis". Global Change Biology. 26 (7): 3821–3833. Bibcode:2020GCBio..26.3821W. doi:10.1111/gcb.15127. PMID 32436328. S2CID 218765595.
- Oehlmann J, Schulte-Oehlmann U, Kloas W, Jagnytsch O, Lutz I, Kusk KO, Wollenberger L, Santos EM, Paull GC, Van Look KJ, Tyler CR (2009). "A critical analysis of the biological impacts of plasticizers on wildlife". Philosophical Transactions of the Royal Society B: Biological Sciences. 364 (1526): 2047–62. doi:10.1098/rstb.2008.0242. PMC 2873012. PMID 19528055.